Volume 3, Issue 1
January 2023
Inflammation Process, Outcome and Prevention of Post-Traumatic Osteoarthritis
Khalid Sunbol, Bashier Al aldahan, Hussain Al Mubarak, Zainab Bozaid, Ibrahem Alqahtani, Abdullah Basarwan, Albaraa Alabbadi, Turki Alghamdi, Hatim Akkasi, Husain Redha, Hisham Almassaud
DOI: http://dx.doi.org/10.52533/JOHS.2023.30104
Keywords: osteoarthritis, posttraumatic, prevent, inflammation
Osteoarthritis is a prominent cause of disability with rising prevalence and incidence. An essential risk factor for posttraumatic osteoarthritis, joint injury history has a substantial role in the rapidly rising prevalence of osteoarthritis in the general population. A prolonged trauma associated with recurring instability leads to the development of posttraumatic oesteoarthritis, which commonly affects the knee and ankle joints. Due to the high number of knee and ankle injuries that occur each year and the strong association between joint damage and the development of osteoarthritis, posttraumatic osteoarthritis represents a significant public health burden. The onset of the chronic disease seems to be significantly influenced by the activation of inflammatory pathways during the posttraumatic osteoarthritis acute phase. Immediately following joint trauma, a number of inflammatory mediators are secreted in synovial fluid. These inflammatory mediators are the predictors of the disease. The purpose of this research is to review the available information about inflammation process, outcome and prevention of post-traumatic osteoarthritis. The continued activation of innate inflammatory pathways may contribute to the maintenance of inflammation and the development of posttraumatic osteoarthritis in the injured joint. Various studies reported more complications observed in posttraumatic osteoarthritis patients as compared to others after knee arthroplasties. Prevention of posttraumatic osteoarthritis shall be targeted before the initiation of clinical onset of posttraumatic osteoarthritis. Anti-inflammatory drugs with sustained delivery can be more beneficial in prevention of cartilage degeneration although further research is needed to study and develop preventive strategies for posttraumatic osteoarthritis.
Introduction
One of the most common debilitating joint conditions, osteoarthritis (OA) causes harm to the articular cartilage, subchondral bone, and synovium in addition to other joint tissues. With considerable effects on those affected, healthcare systems, and large socioeconomic consequences, OA is a major public health concern. Even while OA can affect any joint, it is primarily reported in hip and knee, which, in its latter stages, can result in joint failure and necessitate surgical replacement. Women are more likely than males to develop OA as they age, especially beyond the age of 50. Around the age of 80, the incidence of all joint sites levels off or declines (1). In elderly persons, OA is a major source of impairment and socioeconomic expense. This syndrome is becoming even more common than in earlier decades due to an aging and increasingly obese population (2). Globally, prevalent cases of OA increased by 113.25%, from 247.51 million in 1990 to 527.81 million in 2019. Age standardized ratios were 6,173.38 per 100,000 in 1990 and 6,348.25 per 100,000 in 2019, with an average annual increase of 0.12% (3).
Posttraumatic OA, which typically affects the knee and ankle joints, develops as a result of persistent trauma linked to recurrent instability. A fracture, cartilage damage, acute ligament sprain, chronic ligamentous instability, or a combination of these may be the result of injury. Joint damage changes the biomechanics and neuromuscular control around the injured joint, which may hasten the deterioration of the cartilage. Posttraumatic OA poses a considerable public health burden due to the high number of knee and ankle injuries that happen each year and the close correlation between joint injury and the development of OA. There is limited epidemiologic data on posttraumatic OA. For instance, about 2% of all hip OA cases are related to posttraumatic hip OA. Military members are more likely to have hip posttraumatic OA, with rates reaching 20%. Patients scheduled to undergo a variety of surgical stabilization techniques for anterior glenohumeral instability have posttraumatic OA prevalence at the shoulder that ranges from 8% to 20% (4). ACL injury, meniscus rupture, glenohumeral instability, patellar dislocation, and ankle instability are the five major risk factors that lead to posttraumatic OA (5).
An important aspect of OA is inflammation, which is linked to joint symptoms and the development of the illness. Joint fluids and tissues from patients who have joint injuries and are at risk for developing post-traumatic OA can show signs of inflammation. Additionally, it is believed that inflammatory pathways raise the possibility of OA development and progression following injury. Early on after a joint injury, there is a strong inflammatory response that persists, but it is at a lesser intensity. Although this early inflammatory response, which includes cartilage erosion, contributes to the development of posttraumatic OA characteristics and may be controllable, certain mediators may also be involved in tissue repair (6). The purpose of this research is to review the available information about inflammation process, outcome and prevention of post-traumatic OA.
Methodology
This study is based on a comprehensive literature search conducted on September 26, 2022, in the Medline and Cochrane databases, utilizing the medical topic headings (MeSH) and a combination of all available related terms, according to the database. To prevent missing any possible research, a manual search for publications was conducted through Google Scholar, using the reference lists of the previously listed papers as a starting point. We looked for valuable information in papers that discussed the information about inflammation process, outcome and prevention of post-traumatic OA. There were no restrictions on date, language, participant age, or type of publication.
Discussion
Despite the many advantages, participation in sports and exercise is a major contributor to lower limb musculoskeletal injuries. These injuries have a number of detrimental effects, such as a considerably increased likelihood of developing posttraumatic OA, later comorbidities, and a lower quality of life in terms of health. There is little information on how to stop or delay the onset of posttraumatic OA after an injury has occurred, despite the fact that there is a lot of understanding about how to prevent sports and exercise related musculoskeletal injuries and how to lessen disability in people with posttraumatic OA. Up to 40% of injuries requiring medical attention are caused by sports and recreational activities, and 50% of these injuries involve the ankle, knee, or hip. The ankle and knee are the joints that suffer sports-related injuries the most frequently. Sports and exercise related injuries have a long-term link to obesity, a decline in quality of life, and posttraumatic OA. Within three to ten years after sustaining a sports-related knee injury, young people show greater total body and abdominal obesity compared to uninjured controls (7).
Explanation of inflammation process from literature
There is growing evidence that inflammation plays a critical role in the development of posttraumatic OA. Hemarthrosis, which typically develops in injured knees, may exacerbate inflammation in posttraumatic OA. Joint cavity molecular patterns associated with injury are released by damaged joint tissues. Damage associated molecular proteins, such as uric acid, S-100 proteins, and high-mobility group box protein 1, are produced by dying cells or the extracellular matrix. Damage associated molecular proteins bind to pattern-recognition receptors on the surface of immune cells, chondrocytes, osteoblasts, macrophages, and fibroblasts, as well as injury-toll-like receptors, NOD-like receptors, and the receptor for advanced glycation end-products. Synovial cells release inflammatory cytokines such inter leukines (IL)-1, IL-4, IL-6, IL-8, IL-10, IL-17, and tumour necrosis factor, which are present in higher amounts in injured joints, as a result of this binding activating downstream signalling cascades. Inflammatory chemokines, cathepsins, and complement cascade components are also produced by synovial cells (8).
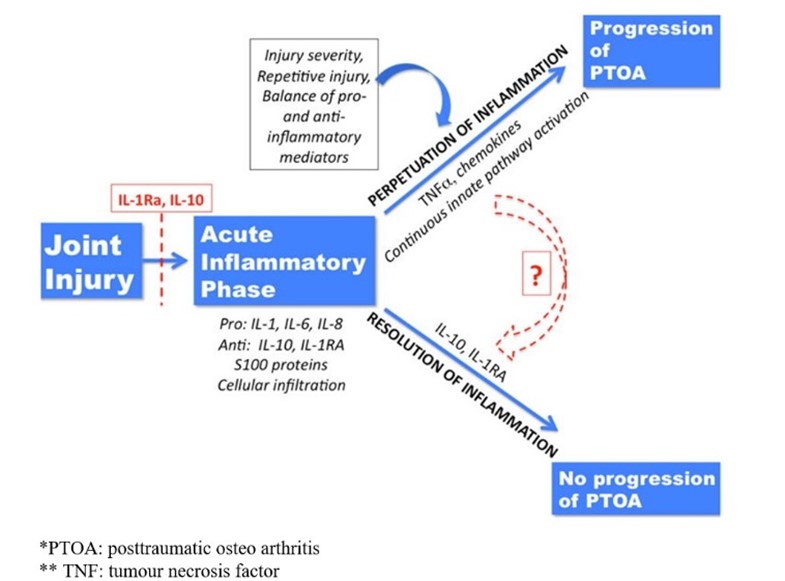
The release of both pro- and anti-inflammatory cytokines, as well as the activation of the complement cascade and other molecular danger signals, are all brought on by joint injury. Anti-inflammatory factors such IL-10 and IL-1RA released in the early post-injury phase may have a positive effect on some people, allowing for the resolution of inflammation and lowering the likelihood of developing posttraumatic OA. However, the development of posttraumatic OA in the damaged joint may be aided by the maintenance of inflammation through ongoing activation of innate inflammatory pathways. After joint injury, a variety of causes might cause inflammation to persist. These factors include the initial injury's severity, the ratio of pro- and anti-inflammatory factors produced in the acute and subacute post-injury periods, and repeated macro or micro injuries that can encourage ongoing cycles of inflammatory activation involving innate pathways like the complement cascade and danger-signal mediated pathways (6). Perpetuation of inflammation is illustrated in (Figure 1).
The joint with OA has been compared to a chronic wound because the development of chronic inflammation in OA after joint trauma or overuse can be regarded as a vicious, self-perpetuating cycle of local tissue damage, inflammation, and healing. A local pathologic inflammatory response is triggered by initial injury, whether it be acute, subacute, or chronic, and it leads to additional cartilage loss and increasing joint deterioration over time. Since chronic damage from previous mechanical derangement such a meniscal rupture or extrusion, overuse, hypermobility, or anatomical misalignment can produce persistent low-grade damage, biomechanics is crucial to the development of OA. However, mechanical derangement is the initiating component in the current paradigm of chronic inflammation. This mechanism is schematically represented in (Figure 2) (9).

Figure 1: Perpetuation of inflammation (6)

Figure 2: Schematic representation of mechanism of chronic inflammation (9)
Thomas et al. stated in his study that despite the fact that OA is typically categorized as a non-inflammatory arthritis, biomechanical stresses affecting the articular cartilage and subchondral bone as well as biochemical changes in the articular cartilage and synovial membrane are significant in its pathogenesis and may be connected to low grade inflammation. Substantial studies indicate that synovium-mediated inflammatory cytokines may distinguish between idiopathic and post-traumatic OA by accelerating disease progression in the latter. Post-traumatic OA's pathophysiology may be synovium-mediated, and it does not always develop along a pathway associated to SDF-1. Future research is required to evaluate additional synovitis-related inflammatory pathways that may initiate and maintain post-traumatic OA and to assist distinguish its pathogenic etiology from idiopathic OA (10).
Outcomes
Bala et al. reported in his study findings that posttraumatic OA patients had a greater incidence of knee wound complications, total knee arthroplasty revision, and arthrotomy/incision and drainage. They also had a higher incidence of periprosthetic infection, cellulitis, or seroma. Patients with posttraumatic arthritis had a reduced rate of blood transfusions. Although these patients are as healthy as those having total knee arthroplasty for primary OA, they are more likely to experience some postoperative surgical complications (11). There are particular technical challenges and complications with total knee arthroplasty as a treatment for advanced post-traumatic arthritis. Technical challenges that surgeons must overcome include old scars, a potential history of infection, malunion-related misalignment, stiffness, and occasionally ligament imbalance. Total knee arthroplasty is therefore more difficult to execute in posttraumatic arthritis than in primary OA (12).
A diagnosis of OA is made in about 12% of individuals who come within 5 years of an anterior cruciate ligament reconstruction. Age, sex, body mass index, tobacco usage, and concurrent meniscal surgery are demographic characteristics linked to a higher chance of receiving a diagnosis of posttraumatic OA within 5 years post anterior cruciate ligament reconstruction. Clinicians should be aware of these risk variables in order to create risk profiles for patients with the shared objective of achieving the best long-term results following anterior cruciate ligament reconstruction (13). Hepp et al. concluded in his study that in comparison to patients with degenerative osteoarthritis, those who have total knee arthroplasty due to posttraumatic OA had inferior outcomes in terms of function and condition. The intricacy of this patient population is highlighted by heterogeneous etiology and high rates of revision surgeries. Both the patient's needs and the pathoanatomy must be taken into account while modifying the course of treatment (14).
Total knee arthroplasty for posttraumatic arthritis affects the pain and functional outcomes depending on the location of the posttraumatic deformity and the soft-tissue envelope impairment. Patients with isolated articular abnormalities, as opposed to those with mixed tibial and femoral deformities, as well as those with soft-tissue compromise, experience much worse outcomes in terms of pain and function (15). Kester.at.al reported that more surgical sites infections, increased duration of intraoperative time, increased demand for transfusion products, and higher readmission rates after total knee arthroplasty were observed in patients of posttraumatic OA (16). Results of a comparative study showed that for OA spurred on by a tibial plateau fracture, lateral unicompartmental knee arthroplasty was effective in regaining joint function, enhancing clinical outcomes, and adjusting lower limb alignment. Clinical results and nine-year survival for primary OA were comparable to lateral unicompartmental knee arthroplasty. Therefore, in carefully chosen individuals, lateral unicompartmental knee arthroplasty should be seen of as a viable alternative for treating lateral posttraumatic OA (17). Findings from a systematic review depicted that despite the challenges of conducting total hip arthroplasty on patients who have posttraumatic arthritis from a prior acetabular fracture and the relatively high complication rates, total hip arthroplasty on patients who have posttraumatic arthritis from a prior acetabular fracture significantly reduces pain and improves function at a 10-year follow-up (18).
Prevention
It is clear that early measures to prevent knee posttraumatic OA are required far before clinical posttraumatic OA onset and eventual disability since posttraumatic OA affects younger individuals than other types of OA. Using various sustained administration techniques, a few studies have examined the immediate removal of corticosteroids and immunoregulators and their brief combined residence times. The development of these various anti-inflammatory sustained delivery systems is encouraging and may enhance the beneficial effects, particularly given that the evidence suggest that sustained delivery approaches are more beneficial than the corresponding single or multiple consecutive treatments because they can control inflammation and cartilage degeneration over a longer time period. This implies that the early posttraumatic OA pathology may be better or more effectively controlled using these therapeutic techniques. The fast clearance from the synovial area can be addressed with a single administration of sustained delivery medication systems.
Repeated injections also prevent injury to the articular cartilage. Even so, very few studies have looked at the use of such strategies for early illness prevention (19). Anakinra after anterior cruciate ligament rupture and dexamethasone after radiocarpal injury have both been studied in human trials for the prevention of posttraumatic OA. With posttraumatic OA, there is a rare opportunity to identify early OA processes that can be therapeutically targeted. Although, this discipline will advance through improvements in trial design, high-calibre preclinical research, and a commitment to patients, regulatory agencies, and the pharmaceutical industry (20).
Anderson.et.al suggested that establishing clinical registries to follow patients with articular injuries over time would allow for the identification of risk factors for the development of posttraumatic OA as well as the expansion of the disease burden. To identify the function of patient characteristics in posttraumatic OA, such as body mass, muscle strength, genetic variations, and activity levels, clinical studies are required. Finally, clinical studies must be planned to be finished in a relatively short amount of time because posttraumatic OA may not manifest for several years following joint injury. The period of time it takes for clinically detectable OA to develop varies greatly; it can take as little as two to five years in the case of some articular fractures. Longer-term injuries may necessitate the continued development of biomarkers that can predict the likelihood of OA, assessments of the severity of joint injuries, early joint healing, and other treatment options (21). Early posttraumatic OA progression prevention becomes an essential therapy, especially for young patients. Recent research has shown that activated fibroblast-like synoviocytes may emit cytokines and enzymes that further erode the cartilage around them. These include interleukin, matrix metalloproteinases, and tumour necrosis factor. While hyaluronic acid with a low molecular weight is more effective at upregulating anti-catabolic enzymes like tissue inhibitor of metalloproteinase-1 and tissue inhibitor of metalloproteinase-2 and at suppressing the catabolic enzyme matrix metalloproteinase-3, which is thought to be more chondroprotective, high molecular weight hyaluronic acid is more effective at downregulating pro-inflammatory cytokines (22). There is a dire need of further research studying the role of inflammation and also focusing on developing therapeutic and preventive strategies for posttraumatic OA as the available literature is quite limited.
Conclusion
Joint trauma commonly leads to posttraumatic OA. It may develop as a result of early post-injury inflammatory processes, such as an increase in inflammatory cytokine release. An effective strategy for lowering or prevention may be anti-inflammatory medication, particularly intra-articular cytokine suppression. However, additional research is required to pinpoint precise biomarkers for the early diagnosis of disease development and to investigate novel approaches to chronic disease prevention.
Disclosure
Conflict of interest
There is no conflict of interest
Funding
No funding
Ethical consideration
Non applicable
Data availability
Data that support the findings of this study are embedded within the manuscript.
Author contribution
All authors contributed to conceptualizing, data drafting, collection and final writing of the manuscript.